How To Draw Contributing Resonance Structures
The Four Key Factors In Evaluating Resonance Structures
Not all resonance forms are of equal importance. And then how do nosotros evaluate how "of import" each resonance construction is?
As we've seen in previous posts, four key factors that determine the importance of resonance structures in organic chemistry are:
- Dominion #1: Minimize charges
- Dominion #ii: Full octets are favored
- Rule #3: How stable are the negative charges?
- Rule #four: How stable are the positive charges?
Today, let's summarize everything we've learned well-nigh resonance structures in this unit.
Table of Contents
- Call up The Three "Legal" Electron-Pushing Arrow "Moves" Used For Interconverting Resonance Structures
- Remember That Resonance Structures Are Not In Equilibrium With Each Other – They Represent Contributions To An Overall Resonance "Hybrid"
- Non All Resonance Forms Are Of Equal Significance. Then How Do We Evaluate How "Important" Each Ane Is?
- Rule #1: Neutral Resonance Structures Are More than "Important" Than Charged Resonance Structures
- Rule #2: Full Octets Are Preferable To Empty Octets (And Never, E'er Have Empty Octets On Oxygen or Nitrogen!)
- Rule #three: Place Negative Charges On The Atom Best Able To Stabilize It (i.e. The Least Bones Atom)
- Rule #4: Place Whatever Empty Octets On the Atoms All-time Able To Stabilize Them (i.e. Carbon And Not Oxygen Or Nitrogen)
- An Awarding Of Resonance: "Pi Donation"
- A 2d Application Of Resonance: "Pi Acceptors"
- Quiz Yourself! (On Evaluating Resonance Structures)
1. Recollect The Three "Legal" Electron-Pushing Pointer "Moves" Used For Interconverting Resonance Structures
Afterwards all these posts about resonance, I thought it would be good to have a post summarizing what'due south been discussed so far.
One of the fundamental skills in analyzing the reactivity of a molecule is to be able to figure out where the electrons are.
As I wrote here, if we're dealing with unmarried bonds, it's a relatively straightforward matter of figuring out the differences in electronegativities.
Notwithstanding if multiple bonds (π bonds) are nowadays, and then nosotros kickoff to run into a piddling problem: at that place tin can be multiple ways to distribute electrons on the same molecule (i.due east. different resonance forms). Therefore, in order to sympathize electron density on a molecule where pi bonds are present, we must first understand the importance of its various resonance forms.
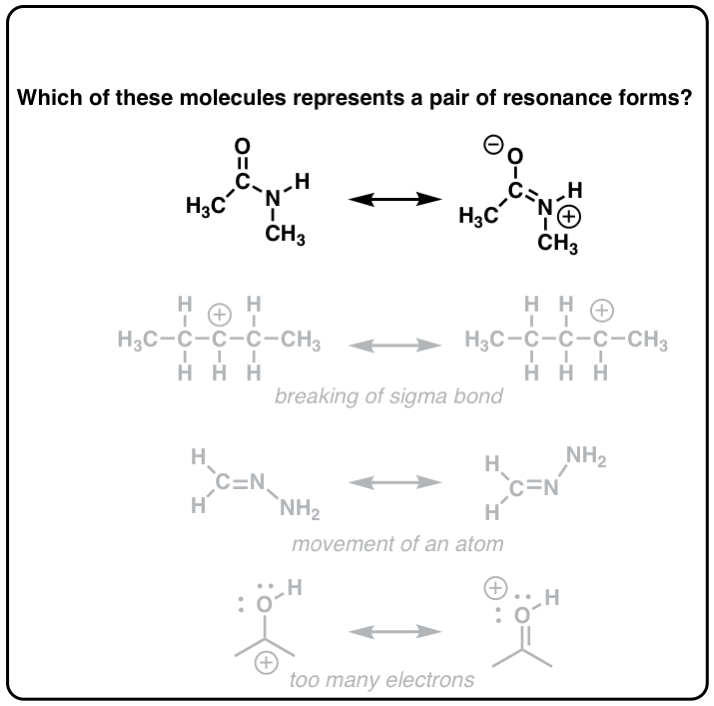
How tin can nosotros "find" resonance forms for a given molecule? It'south possible to do it through trial-and-error, simply one surefire way is to do so is to utilize the curved arrow formalism, which is a way of depicting the "motion" of electrons.
At that place are 3 "legal" ways to motility electrons using curved arrows: from pi bail to lone pair, from solitary pair to pi bail, and from pi bond to pi bond:
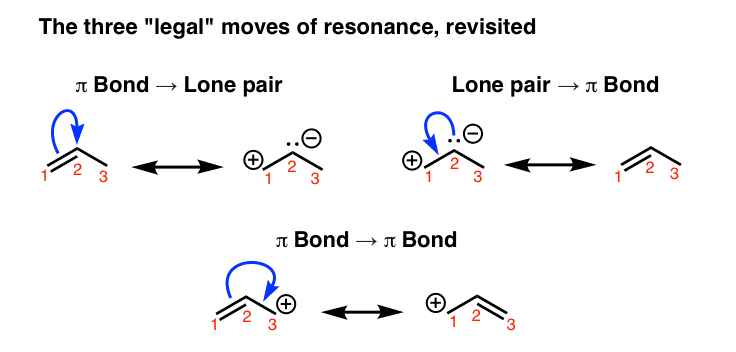
2. Remember That Resonance Structures Are Not In Equilibrium With Each Other – They Represent Contributions To An Overall Resonance "Hybrid"
Here'southward an of import point about resonance forms. It is tempting (and very wrong!) to think that these resonance forms are in "equilibrium" between each other. Avoid this common mistake!
Instead, the "true" land of the molecule will be a "hybrid" of these resonance forms.
For example in the acetate and allyl cation examples below, the "true" structure of the molecule is represented through a fifty:50 combination of the two resonance forms.
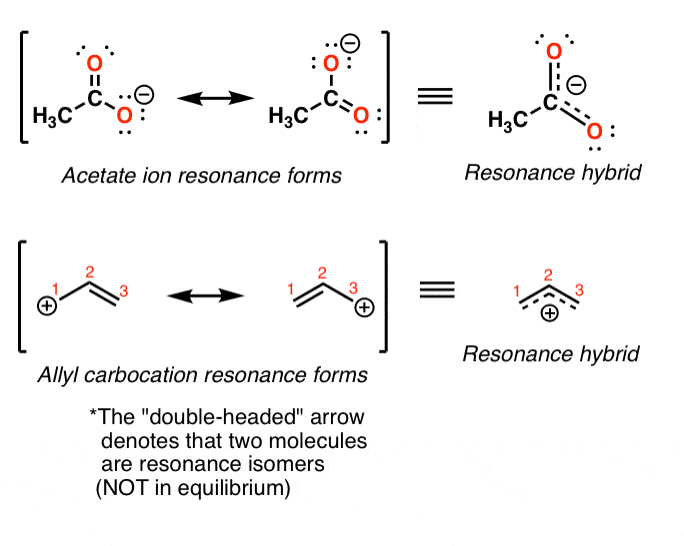
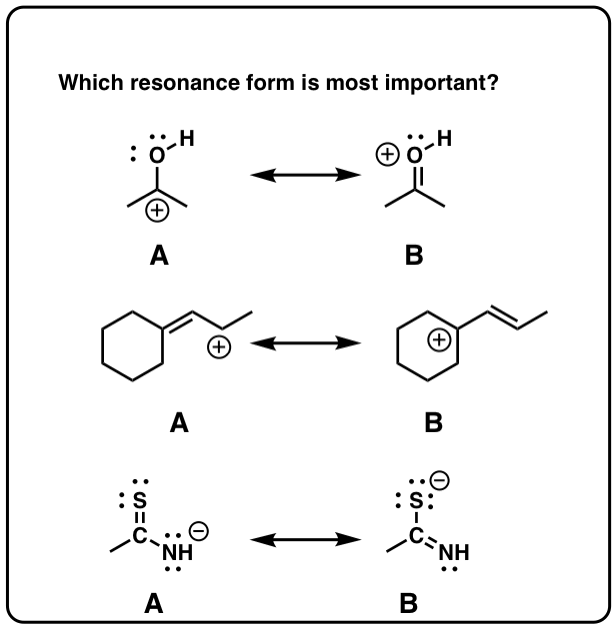
3. Non All Resonance Forms Are Of Equal Significance. And so How Do We Evaluate How "Important" Each I Is?
In the case of the acetate ion and the allyl cation, both resonance forms are equal in energy, so the "hybrid" is a 1:1 mixture of the 2. However, this is only rarely the example.
Accept the ketone below (acetone, or "propanone") for which nosotros can describe iii unlike resonance forms.
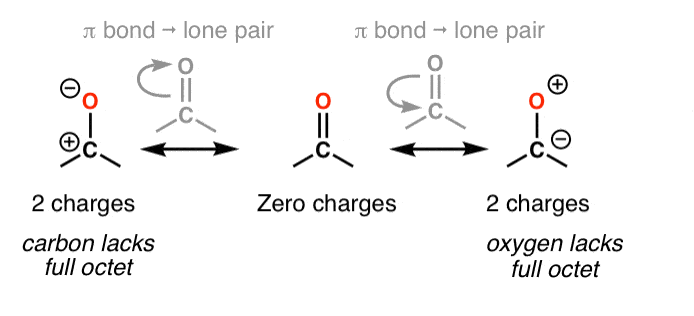
In cases like these, how practise nosotros evaluate the relative importance of each resonance form?
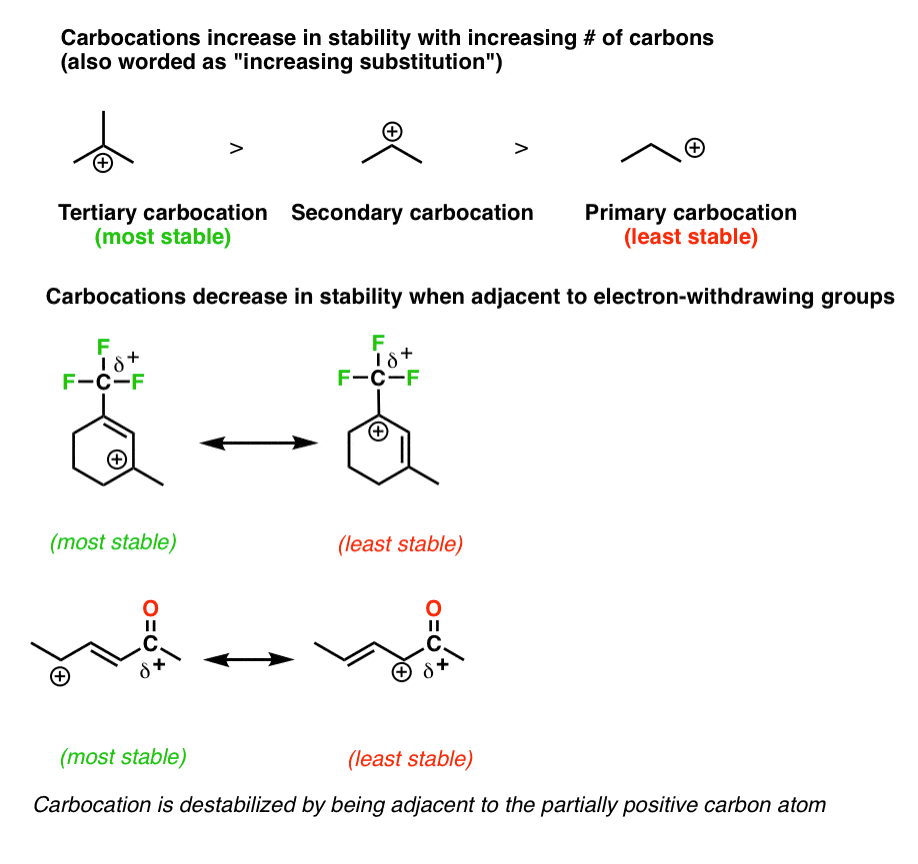
4. Rule #one: Neutral Resonance Structures Are More than "Important" Than Charged Resonance Structures
Resonance forms get less significant as the number of charges are increased (see earlier post).
For example, in the ketone higher up, the resonance course with naught formal charges will exist the nigh significant.
How practise we know? Nosotros can measure out the concrete properties of the molecule (e.grand. humid points, solvent properties, conductivity) and meet if information technology'southward more consistent with a charged species or a neutral chemical compound.
All the physical properties of propanone (acetone) are consistent with it existence a (mostly) neutral molecule. For example acetone has a boiling bespeak of 56°C, significantly lower than water, and a freezing signal of –95°C. It doesn't deliquesce charged species (like NaCl) nearly as well as h2o does. And it isn't a particularly good conductor of electricity.
That isn't to say that the "second-all-time" resonance form doesn't play some part. Acetone is much higher-boiling than butane (–1°C) which has a similar molecular weight due to the dipole-dipole Van der Waals attractive forces, and every bit nosotros'll see afterward, the "second-all-time" resonance course tin yield an important clue as to the reactivity of a molecule.
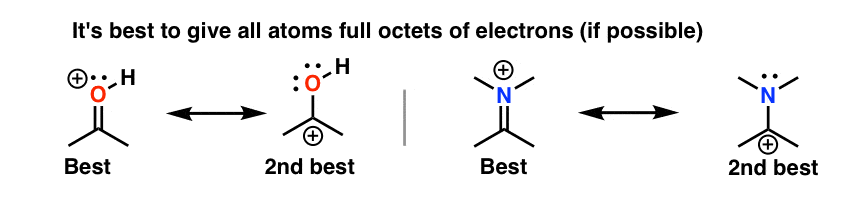
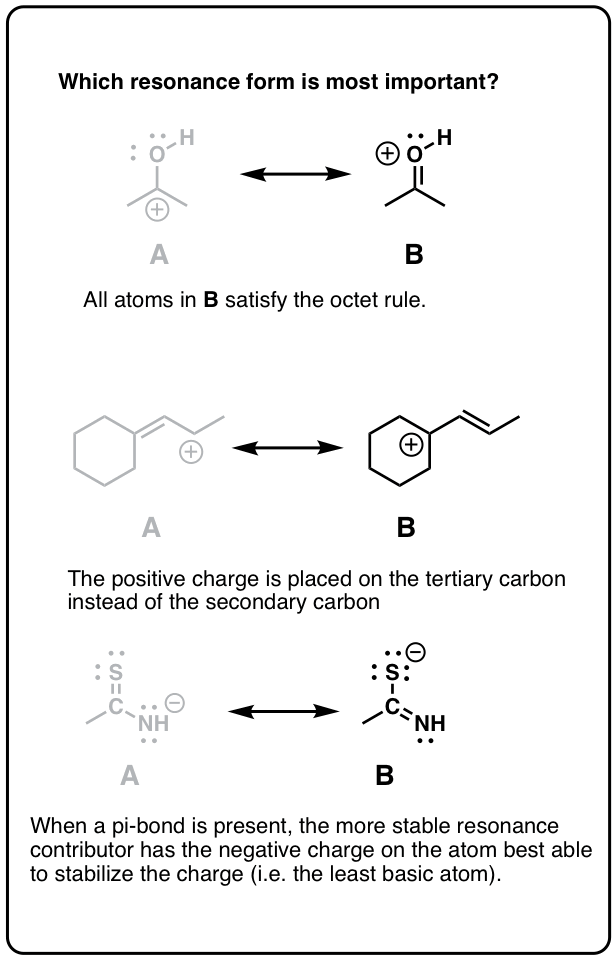
5. Dominion #2: Total Octets Are Preferable To Empty Octets (And Never, Ever Take Empty Octets On Oxygen or Nitrogen!)
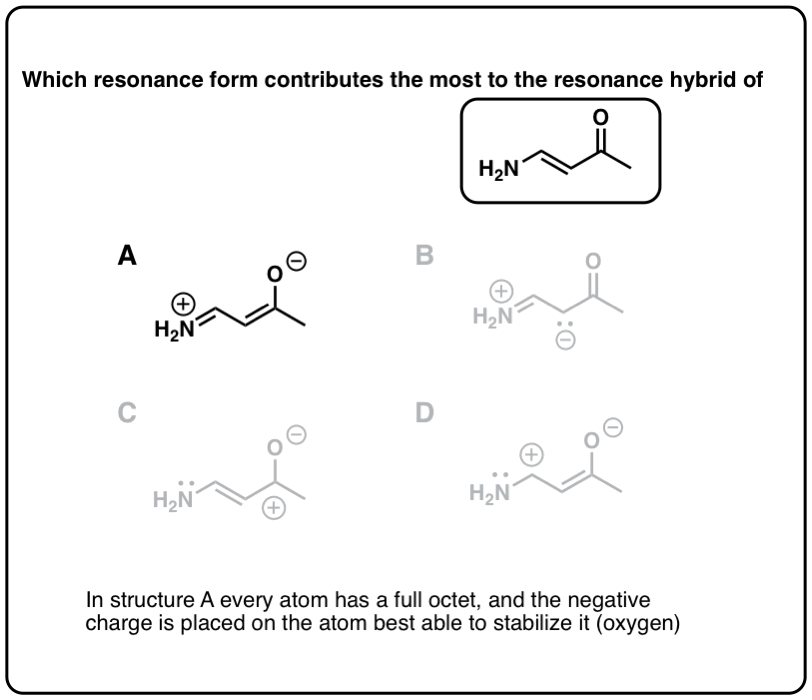
Resonance forms where all atoms have full octets will be more pregnant than resonance forms where atom(s) lack a full octet. Importantly, it'south a good general rule never to identify less than a full octet on nitrogen or oxygen, as in the acetone example (above right). Since these atoms are highly electronegative, these resonance forms are extremely unstable and will be insignificant.
half dozen. Rule #iii: Place Negative Charges On The Atom Best Able To Stabilize It (i.eastward. The To the lowest degree Basic Atom)
Given that neutral resonance structures are preferred overall, when a resonance construction admittedly must comport a negative charge somewhere, place it on the cantlet best able to stabilize that charge. Since, in essence, "basicity is the reverse of stability", this is the same equally saying, "put the negative charge on the least bones atom".
The good news hither is that if y'all empathise the factors that affect acerbity, you also empathise the factors which stabilize negative charge.
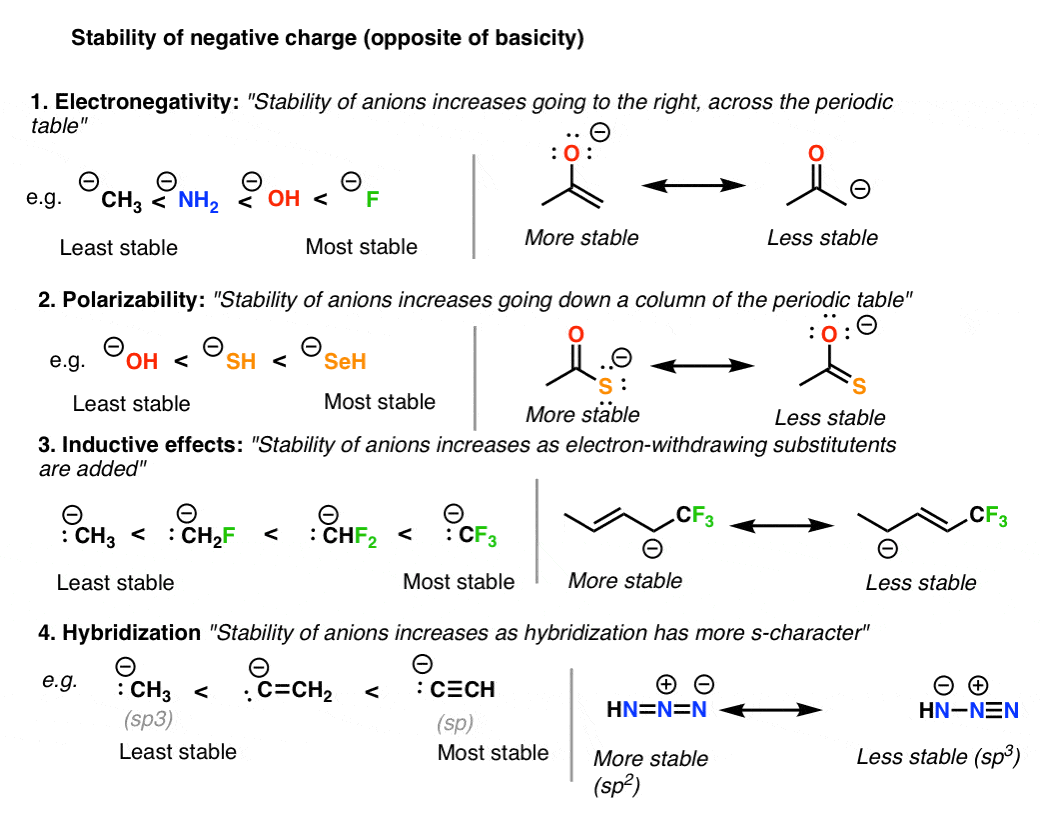
There are 4 main trends to consider here:
- Electronegativity: beyond a row of the periodic table, negative charge becomes more stable every bit electronegativity is increased.
- Polarizability: down a cavalcade of the periodic table, negative charge becomes more stable as polarizability increases
- Electron withdrawing groups stabilize negative charge through inductive furnishings.
- Hybridization: negative charge becomes more stable as the south-character of the cantlet is increased. sp (almost stable) > sptwo > sp3 (to the lowest degree stable
Note again that stability is the reverse of basicity.
7. Rule #4: Place Any Empty Octets On the Atoms Best Able To Stabilize Them (i.e. Carbon And Not Oxygen Or Nitrogen)
As we said above, full octets are best. Even so, when dealing with a resonance structure where there absolutely must exist an cantlet with less than a full octet, then follow these principles:
- Place the empty octet on carbon, never oxygen or nitrogen
- Place place the empty octet on the nigh substituted carbon(think carbocation stability)
- Avoid placing positive accuse side by side to electron withdrawing groups if possible
- Place positive charge preferentially on alkyl carbocations as opposed toalkenylor (peculiarly)alkynylcarbons.
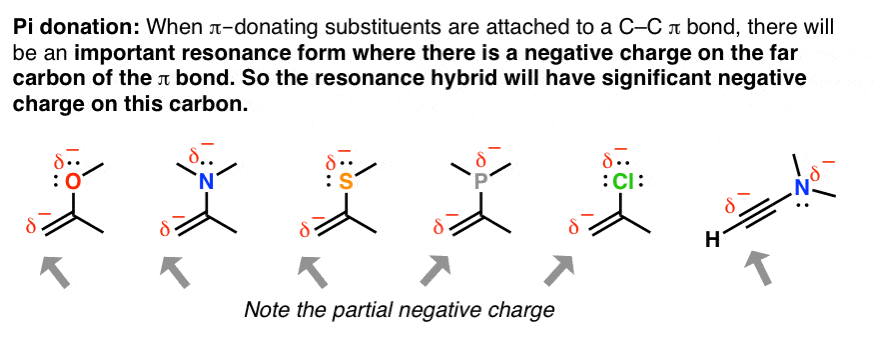
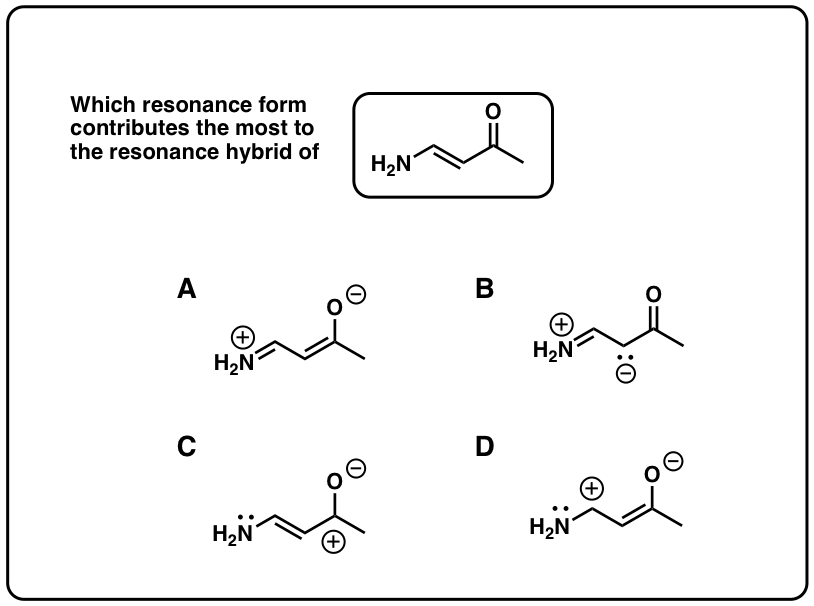
viii. An Awarding Of Resonance: "Pi Donation"
When double bonds are connected to an atom with a solitary pair of electrons, the molecule will have a pregnant resonance form where there is negative charge on the adjacent carbondue to a phenomenon called, "pi donation". This becomes particularly important once you starting time learning almost reactions of pi bonds.
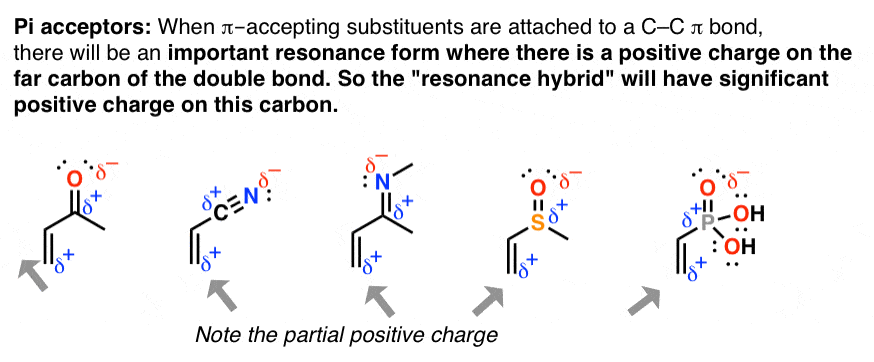
nine. A 2nd Application Of Resonance: "Pi Acceptors"
When double bonds are connected to a polarized π bond, the molecule will accept a significant resonance grade where there is positive charge on the adjacent carbon. This phenomenon is known as "pi-accepting" behavior, and these groups are known as "pi acceptors".
For now, that does it for a summary of the of import themes in resonance. Next stop (later a post about some common mistakes) will be to apply these principles to chemical reactivity.
Next Post: Common Mistakes – How NOT to Draw Resonance Curved Arrows
Quiz Yourself on Resonance Structures
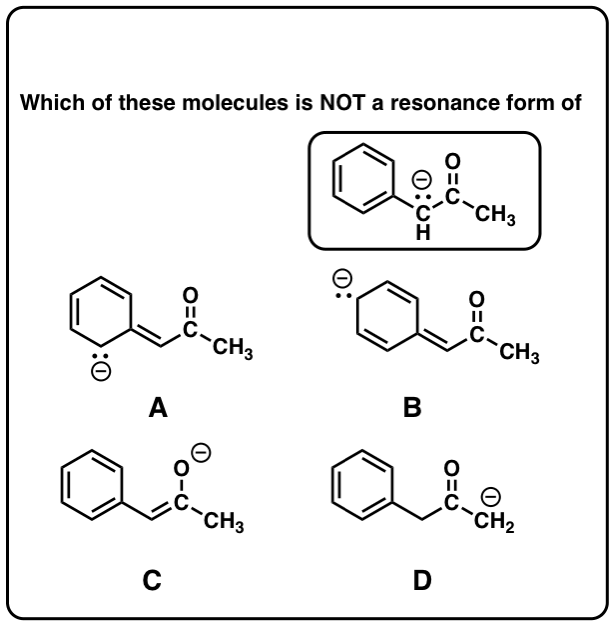 Click to Flip
Click to Flip
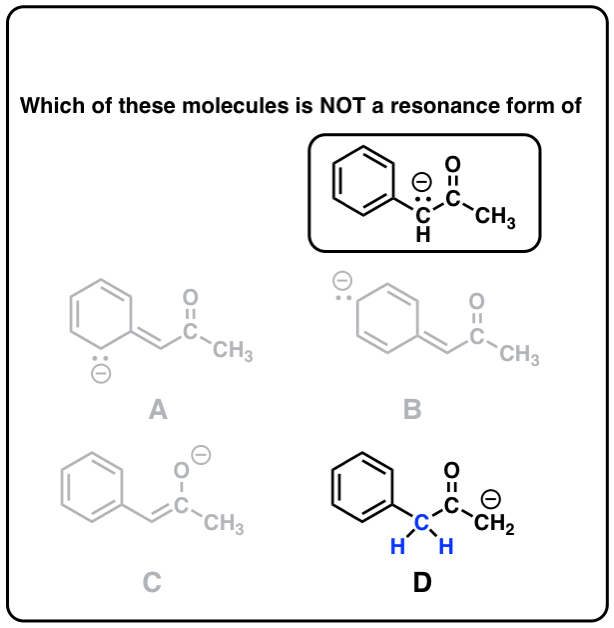
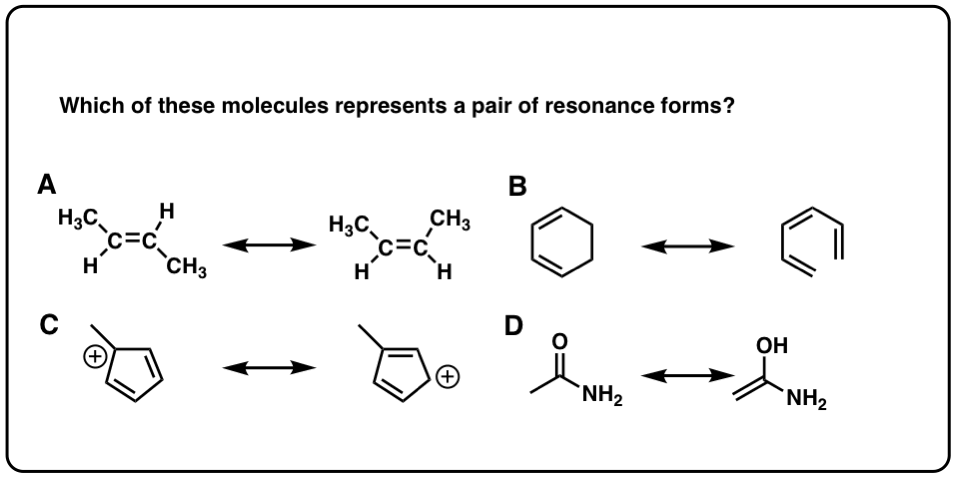 Click to Flip
Click to Flip
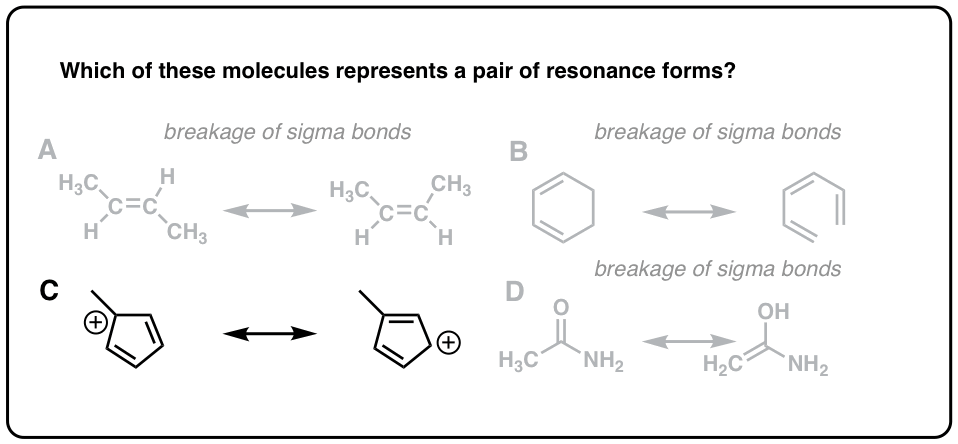
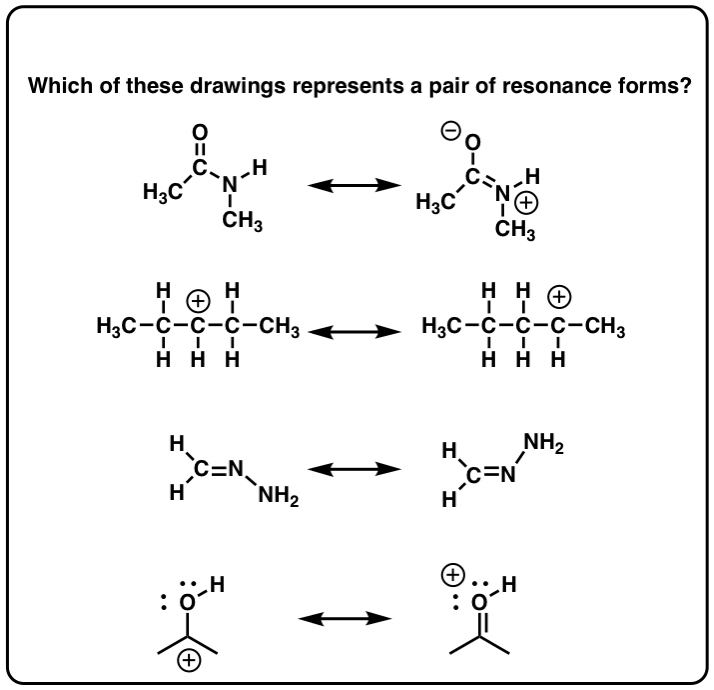 Click to Flip
Click to Flip
 Click to Flip
Click to Flip
 Click to Flip
Click to Flip
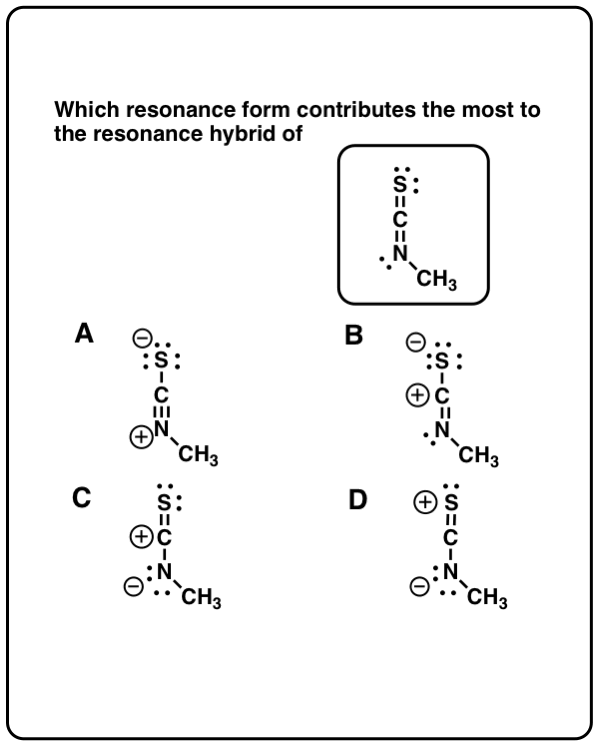 Click to Flip
Click to Flip
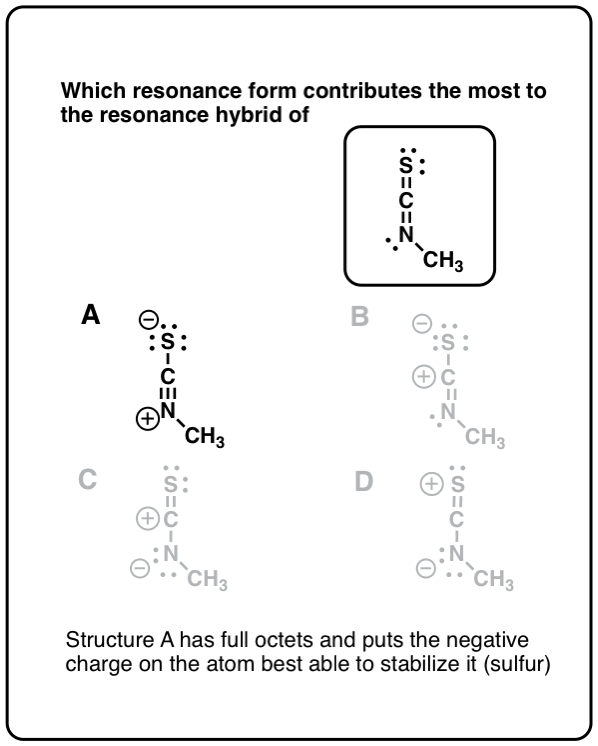
Source: https://www.masterorganicchemistry.com/2011/12/22/in-summary-resonance/
Posted by: claussenmades1969.blogspot.com


0 Response to "How To Draw Contributing Resonance Structures"
Post a Comment